Publication: Biosensor for Lyme disease: real-time, specific, and label-free CMOS transistor sensing of Borrelia burgdorferi s.l. outer surface protein A in unprocessed blood
Journal Link: Click here.
Authors: Vijay Garika 1, Vladimir V. Bamm 2, Pooja Verma 1, Shubham Babbar 1, Surbhi Rajpoot 1, Shankar Bhattarai 3, Sherina Harilal 4, Ayat Asleh 4, Inna Shehter 5, Ayala Elkayam 5, Iain L. Mainprize 2, David Akselrod 2, Avital Eisenberg-Lerner 6, Ziv Rotfogel 6,7, Evgeny Pikhay 5, Muhammad Y. Bashouti 4,8, Barak Akabayov 3, Izhar Ron 1, Yakov Roizin 5, Melanie K. B. Wills 2, Gil Shalev 1,8*
Affiliations:
1 School of Electrical Engineering, Ben-Gurion University of the Negev, Beer-Sheva 8410501, Israel.
2 G. Magnotta Lyme Disease Research Lab, Department of Molecular and Cellular Biology, University of Guelph, Guelph, Ontario N1G 2W1, Canada
3 Department of Chemistry and Data Science Research Center, Ben-Gurion University of the Negev, Beer-Sheva 8410501, Israel.
4 Department of Solar Energy and Environmental Physics, Swiss Institute for Dryland Environmental and Energy Research, J. Blaustein Institutes for Desert Research, Ben-Gurion University of the Negev, Midreshet Ben-Gurion 8499000, Israel.
5 Tower Semiconductor, Migdal Haemek 2310502, Israel
6 Ophthalmology Research Laboratory, Kaplan Medical Center, Rehovot 7661041, Israel.
7 Faculty of Medicine, Hadassah Medical School, The Hebrew University of Jerusalem, Jerusalem 9112001, Israel.
8 The Ilse-Katz Institute for Nanoscale Science and Technology, Ben-Gurion University of the Negev, Beer-Sheva 8410501, Israel.
Short Summary
The diagnosis of Lyme disease remains challenging in the clinic and the medical laboratory, which makes it very hard for doctors and labs to tell if someone has Lyme disease. We need fast and accurate tools to help people get the right diagnosis and proper medicine quickly. Historically the biggest challenge is that the germ that causes Lyme disease (called Borrelia burgdorferi s.l.) hides in the body in such tiny amounts that it is almost impossible to find. Therefore, this problem is often compared to “finding a needle in a haystack”.
Our collaborative research demonstrates that we have created a new kind of sensor that “feels” the presence of even the tiniest amounts of a Lyme disease biomarker using an integrated circuit (or microchip) designed to translate the finding of the biological target into electrical signals that a computer can read.
The MNC-FET sensor (our “smart” microchip), described in more detail below, was found to have the following characteristics:
Superb sensitivity: It can find very low amount of the Lyme biomarker even if it is floating in a huge amount of blood (lower limit of detection was found to be 1 fg/ml of target protein in whole blood).
High specificity: no cross-reactivity detected in our trials, meaning the sensor does not respond to unrelated molecules. In other words, the sensor won’t give a “false alarm”.
Small sample volume: The sensor needs only a tiny bit of blood (0.5 ul of whole blood) to detect the protein of interest, which is roughly equivalent to 1/60 of one drop of blood.
Label-free: Usually, blood samples must be “cleaned” or mixed with special dyes in a lab before they can be tested. Our sensor does not need that; the blood sample is applied directly on the chip.
Minimal handling: whole blood is added to the surface of the sensor and measured directly, without washing.
Rapid: sensing happens in real time, with no need to wait hours or days for a lab to call you back.
Consistent manufacturing: the sensing chips are fabricated by a semiconductor factory (Tower Semiconductor) with very strict rules and under stringent quality control for exceptional electronic performance. Next, the chips are coated with a special “sensing” molecule (biofunctionalization) that allows them to recognize only the specific Lyme disease biomarkers.
Point-of-care potential: with further development of this sensor reading technology, the chip could be made into a small device people could use at home, or at the clinic and pharmacy, just like a glucose monitor.
Deeper Perspectives
Why: The diagnostic problem
Laboratory diagnostics are a cornerstone issue in Lyme disease both from a clinical and scientific perspective. Patients and healthcare providers need accurate tools to rapidly understand what is causing illness. Researchers studying the effects of Lyme disease or evaluating new treatments need to clearly identify individuals with a Borrelia infection and distinguish them from those with other lookalike illnesses. The current standard test is not fulfilling these needs.
The Current Strategy: Searching for “Footprints” (Indirect Testing)
Conventional lab testing for Lyme disease is considered indirect. Instead of looking for the actual bacteria (Borrelia), the test looks for immunological evidence (antibodies), or the “footprint” that the host immune system leaves behind after encountering the germ. In this test, whether in the original or modified two-tier testing algorithms, both tiers evaluate host antibodies. There are three major flaws in this test:
The lag time: The immune response that is read by this test is slow to develop. If an individual is tested too early, even if they are sick, the result might come back as “false negative”.
The “Ghost” signal: Antibodies stay in the blood for years, which means the test can’t tell if you have a new infection, a re-infection, or if the previous treatment actually worked.
Immune sabotage: The Lyme disease bacteria (Borrelia) can interfere with the immune system, making it harder to produce the antibodies the test is looking for.
In a climate where the infected tick population is rapidly expanding, human-tick encounters are escalating, and patients can experience long-term debilitating illness even after treatment, tests that measure exposure to the pathogen are simply not adequate to deal with the problem.
The Goal: The “Holy Grail” (Direct Testing)
The “holy grail” of Lyme diagnostics is a direct approach that can detect the pathogen or its unique components (which we will refer to as biomarkers), such as a specific protein or a piece of DNA, in patient samples. In other words, if we can find the bacteria’s “ID card” in the blood, we know for a fact the infection is active.
Historically in clinical microbiology, this has involved culturing the microorganism directly from the site of human colonization (tick bite site). More modern techniques aim to rapidly detect unique proteins or DNA signatures that can identify the disease-causing agent.
These strategies have proven challenging for the Lyme pathogen Borrelia, which is not abundant during infection and disperses throughout the body, colonizing sites distant from the initial tick bite. Borrelia is also difficult to grow in the lab. Consequently, the amount of biological material available to a diagnostic test is often below the threshold of detection. A valid strategy to report the presence of the pathogen needs to be extremely sensitive without compromising specificity. In other words, it needs to find the exact molecule of interest in a very crowed biological space, and not respond to other molecules that share similar features. So, the ideal test resembles “finding a needle in a haystack”.
Who
The potential to use electrochemical platforms to detect Borrelia biomarkers was one of the first concepts we began to explore after the G. Magnotta Lab was established in 2017. Led by biochemist Dr. Vladimir Bamm, with support from microbiologist Dr. Iain Mainprize and Visiting Scientist, engineer Dr. David Akselrod, we initially trialed two different sensor designs in-house. Considerable experimentation and troubleshooting resulted in mixed success, and it became clear that the devices we were creating did not have the properties required to underpin extremely sensitive and accurate diagnostic tests.
Surveying the international landscape of advanced biosensor design, we identified several promising research groups, but none more so than the lab of Dr. Gil Shalev at Ben-Gurion University of the Negev. He and his group had recently introduced their novel Meta-Nano-Channel Field-Effect Transistor (MNC-FET), which offered several advantages over other modalities of biosensing. Dr. Shalev was eager to determine whether his platform would address the biological challenges presented by Lyme disease, so a collaboration was established to modify his sensor using our biomarker capture strategy and test its performance. These experiments were conducted by electrical engineering PhD candidate Vijay Garika. Ultimately, the project involved cross-sector contributions from academia, medicine, and semiconductor manufacturing and the multidisciplinary expertise of the team in engineering, material science, physics, microbiology, immunology, and biochemistry. Work performed by the G. Magnotta Lab at the University of Guelph was supported by the G. Magnotta Foundation for Vector-Borne Diseases and Caron Thorburn Institute.
What: the OspA Biosensor
This collaboration ultimately gave rise to a sensor that overcomes key limitations of Borrelia biology and the technical challenges of biosensing. The platform was designed to specifically and exclusively capture Outer surface protein A (OspA), which is abundant on the exterior of pathogenic Borrelia and also released into its surroundings. It is the same protein on which the Nanotrap Lyme Antigen urine test is based.
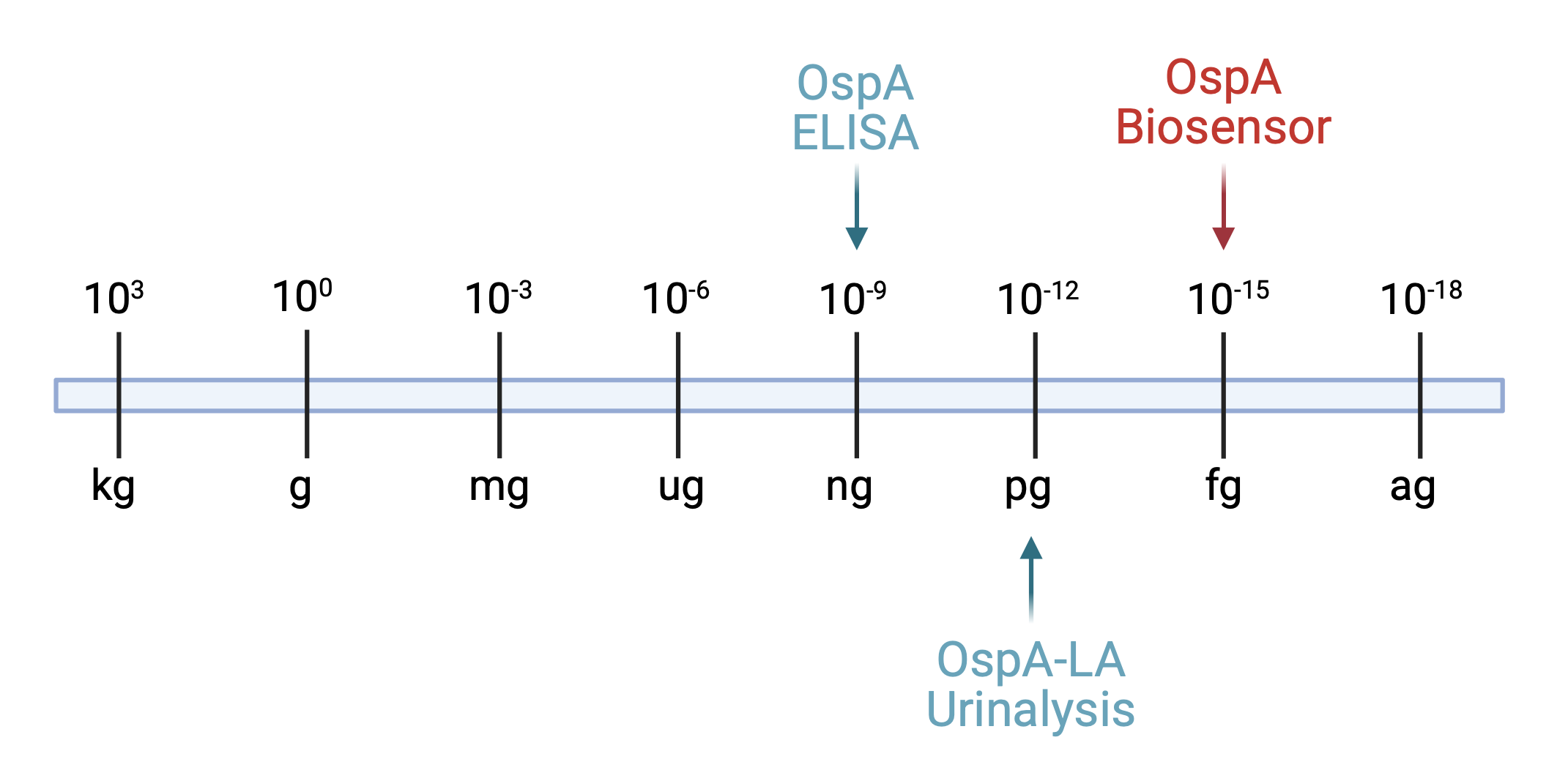
Unlike the Nanotrap design described by Magni et al in 2015, which concentrates OspA using nanoparticles and then detects the output using traditional biochemical methods (Western blot), the biosensor translates molecular recognition into electrical currents. Miniscule amounts of the biomarker bound to the sensor can generate an electrical signal that can be amplified, allowing this platform to be exquisitely sensitive. In figure 1, the scale of biosensor detection is shown relative to a standard protein assay (ELISA), which in our hands achieved a limit of OspA detection of 2.5 ng/ml in human serum (billionths of a gram), and Nanotrap, with its reported LoD of 1.7 pg/ml of urine (trillionths of a gram). Our biosensor can detect amounts 1000x lower (quadrillionths of a gram), with no sample processing required.
Figure 1: Scale of detection. The OspA biosensor analytical lower limit of detection is shown relative to conventional biochemical techniques (ELISA) and the Nanotrap Lyme Antigen test. Figure created in BioRender. Wills, M. (2026) https://BioRender.com/gl22ays
So, the advantage of our biosensor is that, unlike in traditional testing, where samples have to be “prepared” using centrifugation for the removal of the cells or sedimentation of the concentrating nanoparticles, our biosensor uses label-free and direct approach. The raw sample (like a drop of whole blood) is applied directly onto the chip, and it identifies the OspA immediately without any cleaning or enrichment steps and with superior sensitivity.
How
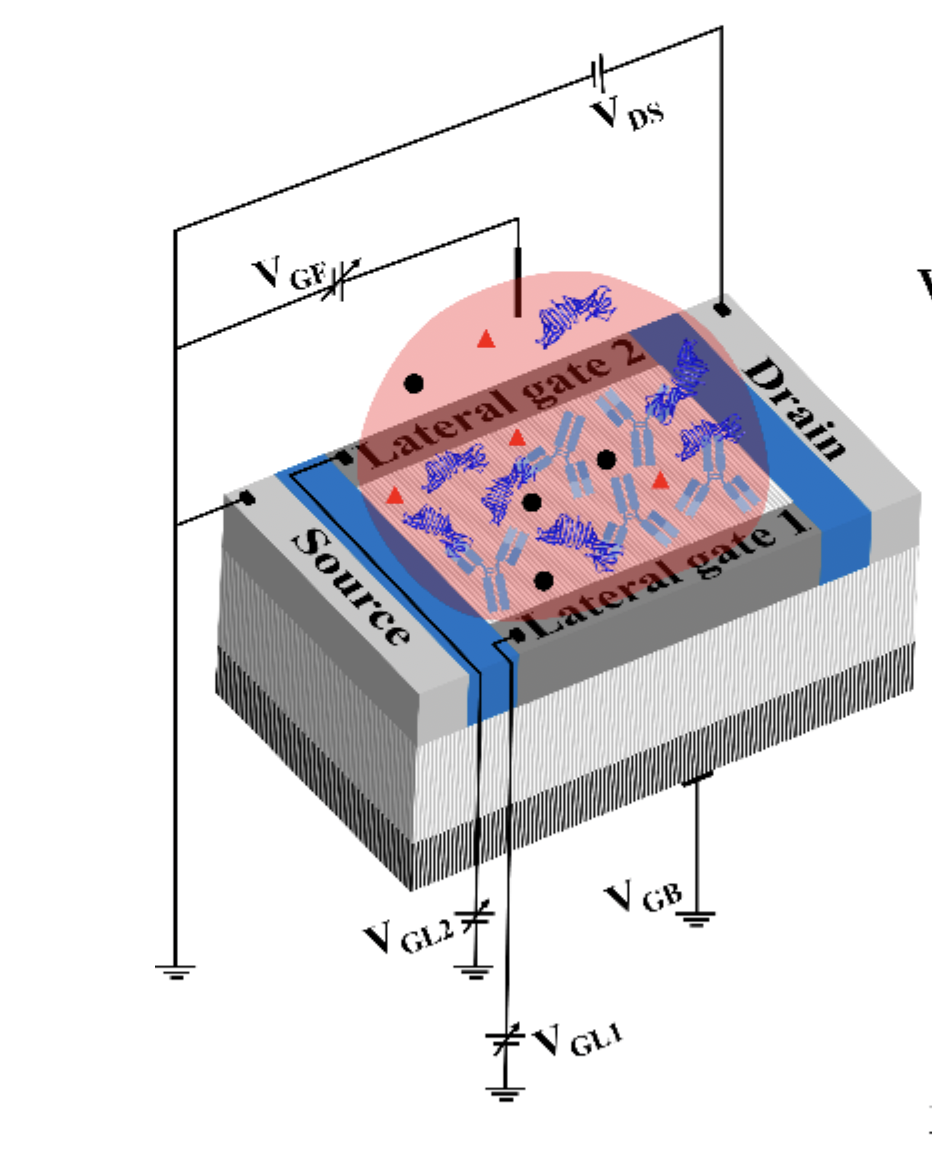
The foundation of this detection platform is a tiny, purpose-designed, precision manufactured integrated circuit fabricated using a Complementary Metal Oxide Semiconductor (CMOS) technology (Figure 2), the same high-standard process used to make the microchips in phones, laptops, and other smart electronic devices. The sensor is then functionalized for this application using a traditional biochemical capture strategy of immobilizing target-specific antibodies on the surface that uniquely recognize and bind our protein of interest, OspA.
Figure 2: schematic representation of the sensor showing the circuit design (source, drain, gates), functionalization (antibodies), and drop of blood containing target (red bubble).
The biosensing circuit consists of numerous narrow channels through which electrons can flow from the source to the drain to create a current that can be measured (the “nano channel” portion of the MNC-FET). Under the electrochemically defined conditions of the analysis, these channels generate negligible signal at baseline. However, when the functionalized sensor captures target molecules (OspA), this creates a non-uniform electrical potential distribution close to the sensing surface (field effect) which in turn generates a signal above baseline (IDS; current; “field effect transistor” of MNC-FET).
Essentially, this biosensor is a biological transistor. It requires the binding of the specific Lyme disease biomarker (OspA) and uses its natural field effect to toggle the current on a chip. This allows us to detect the presence of the disease at a molecular level using electricity rather than slow, traditional lab biochemistry.
The platform was tested in the lab by introducing known amounts of OspA into whole blood. The magnitude of the signal (current, measured in nA) was proportional to the amount of OspA in the sample over a range of 8 orders of magnitude in these trials.
This is significant because Borrelia biomarkers are expected to be present at very low concentration in infected individuals, although they can fluctuate with the stage of the disease. Additionally, whole blood is a complex fluid containing off-target proteins and ions that can make it electrochemically noisy. The capacity to isolate the legitimate pathogen signal from the background noise of the blood is a noteworthy feature of this sensor.
Overall, the design of the sensor and its biological-chemical interface allow for exquisite detection sensitivity that happens in real time with no pre- or post-application processing, making the measurement itself very streamlined.
What’s Next
While this represents a breakthrough in detecting Lyme OspA in the blood, the sensing device is a lab-based prototype that cannot yet be mass produced or deployed at scale. From an engineering standpoint, the components supporting the sensor that record and interpret the measurements are bulky and need to be miniaturized, packaged, and integrated with a straightforward user interface. Biologically, expanding the number and diversity of targets will make the test more robust. By adding different capture antibodies to the chip (multiplexing), the sensor could look for multiple different biomarkers or even different strains of the Borrelia bacteria at the same time. This would act as a double-check system to ensure accuracy across different stages of the infection.
Importantly, it also requires clinical validation and regulatory device trials to establish its performance in a real-world setting. Its performance needs to be tested in early-stage vs. late-stage Lyme disease and during the medical intervention to ensure that it works across all the conditions and populations. There are many factors that will influence whether, and how, the biosensor moves through the next phases of development. With its performance and characteristics, however, it is positioned to solve several problems plaguing Lyme disease diagnostics. The “End Game” for this technology is point-of-care use. Instead of sending blood to a centralised testing facility and waiting weeks for result, our sensor is designed to be used at the point where the patient is cared for, or even at home.
The Big Picture
We have the "engine" (the ultra-sensitive sensor), but we still need to build the "car" (the handheld device), test it on real roads (clinical trials), and get the driver’s license (regulatory approval). Because the sensor is so powerful, it has the potential to finally give us the fast, sensitive, specific, and direct Lyme disease test that has been missing for decades.